Drug Delivery Research Unit
- Establishes a full-cycle performance evaluation system covering design, manufacturing, analysis, and validation of targeted bionano drug delivery systems and antibody–drug conjugates (ADCs) as part of a precision drug delivery platform
- Identifies and functionally validates target genes that define the pathogenic mechanisms of cancer and aging-related diseases, thereby simultaneously optimizing therapeutic targets and delivery modalities
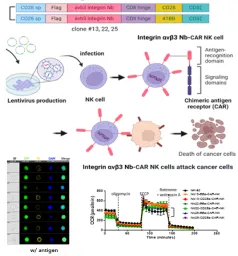
- Develops immune cell–based anticancer cell therapies and technologies for functional enhancement
- Establishes a preclinical efficacy evaluation system linking 2D cell / 3D organoid–based in vitro models with in vivo animal models
Research Content
Research Infrastructure
Analytical Infrastructure of the Drug Delivery Research Unit
| Equipment / Service Name | Person in Charge (Primary/Secondary) | Year Installed |
|---|---|---|
| Imaging Flow cytometry | Hyesun Park/ Eunhee Han | 2025 |
| IVIS spectrum | Hyunseung Lee/ Hyesun Park | 2017 |
| Cell sorter | Eunhee Han/ Hyesun Park | 2015 |
| Confocal Microscopy | Eunhee Han/ Cheoljoong Lee | 2009 |
- Utilizes the Amnis ImageStream MKII to simultaneously acquire and analyze high-resolution fluorescence images at the single-cell level along with multiparametric flow cytometry data, enabling quantitative characterization of cellular heterogeneity and intracellular uptake, distribution, and transport behavior of nano/bio delivery systems and ADCs
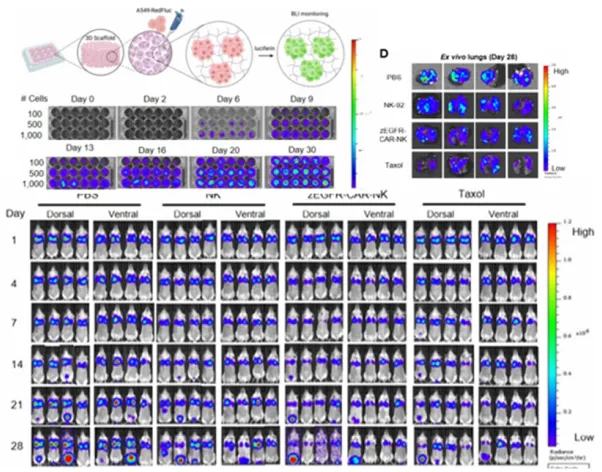
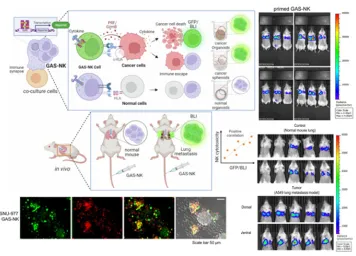
- Utilizes the IVIS Spectrum to non-invasively track the biodistribution, tumor accumulation, drug delivery efficiency, and therapeutic response of nanocarriers, ADCs, and immune cell therapies in animal models, while establishing a preclinical efficacy evaluation system based on in vitro–in vivo correlation (IVIVC) by linking results from 2D cell and 3D organoid in vitro models.
- Uses a high-speed automated cell sorting system(Cell sorter) to isolate immune cell therapeutics such as CAR-T/NK cells with high purity and validate their functions. Tumor- or organoid-derived cells are further sorted into groups based on target expression levels (high/low), resistant subtypes, and immune-evasive subtypes to compare responses to DDS, ADC, and cell therapies, thereby identifying predictive biomarkers and evidence for combination strategies.
- Uses confocal microscopy to quantitatively analyze intracellular entry, spatial distribution, organelle colocalization, endosomal escape, and drug release of targeted bionano delivery systems and ADCs. Visualization of penetration depth, target-binding specificity, and microenvironmental effects (hypoxia/ECM) in 2D cells and 3D organoids enables elucidation of the platform’s mechanism of action (MoA) and efficacy-determining factors, as well as optimization of design parameters (ligands, linkers, payloads, and physicochemical properties).
- Utilizes multi-omics–based target discovery and validation infrastructure (including transcriptomics and proteomics) to systematically identify disease-specific genes, proteins, and pathways related to cancer and aging. By interpreting target expression patterns, heterogeneity among patients and models, and associations with drug response, the unit advances target selection and indication strategies for bionano/ADC platforms and drug candidates.
- Establishes gene regulation–based functional validation systems (e.g., CRISPR/Cas9 and overexpression) together with cell-based functional analyses to precisely verify the pathogenic contribution, vulnerability, and drug sensitivity of candidate targets. Comparative analysis of DDS/ADC delivery efficiency and cell therapy responsiveness upon target modulation enables mechanism-based prioritization of therapeutic candidates.
- Operates an in vivo validation system using animal models to confirm the functional relevance and therapeutic intervention potential of candidate targets. By integrating IVIS-based biodistribution and response data, the unit performs comprehensive preclinical evaluation of efficacy, distribution, and safety of DDS, ADCs, and cell therapies.
- Integrates analyses of expression characteristics (quantitative and spatial), intracellular localization (membrane, cytoplasm, nucleus, organelles), and functional vulnerabilities (synthetic lethality and resistance pathways) of identified targets, thereby establishing a foundation for generating integrated basic-to-preclinical data directly applicable to targeted therapy strategies.
Research on elucidating the in vivo biodistribution of exosomes using near-infrared fluorescence imaging
Journal of Extracellular Vesicles, IF=15.5, JCR 16.47%, ’24.07.
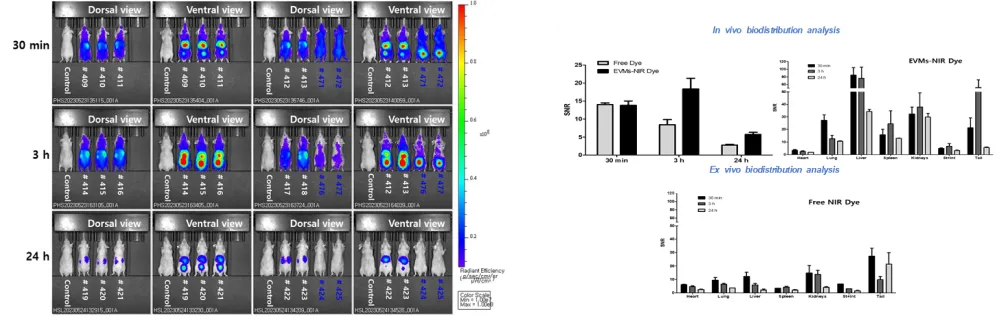
Development and analysis of fluorescent dye–labeled probes for investigating the in vivo distribution of bioactive substances
Journal of Extracellular Vesicles, IF=15.5, JCR 16.47%, ’24.07.
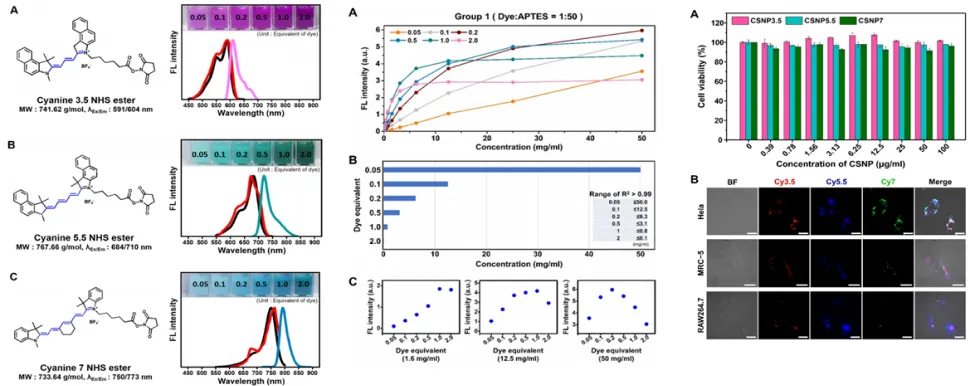
Nanobio drug analysis technology: multimodal efficacy evaluation of gold nanohybrid systems
ACS Appl Mater Interfaces, IF=8.2, JCR=18%, ’25.03.
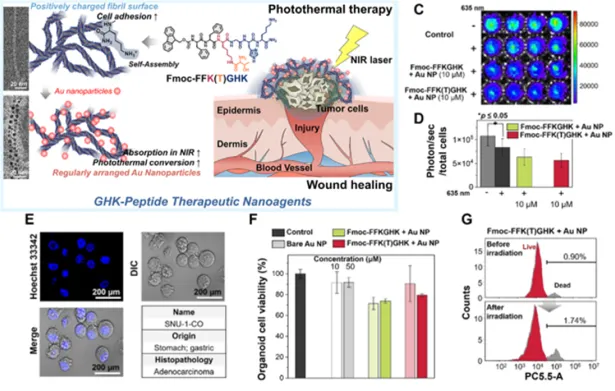
Non-invasive monitoring and efficacy evaluation of NK therapies using 3D models reflecting immune-resistant tumor microenvironments
ACS Applied Bio Materials, IF=4.2, ’24.11.
Communications Biology, IF=5.1, ’24.09.